Our Approach
The Drug Discovery Lab approach is based on the hypothesis that AD results from imbalances in an extensive array of networks. Therefore, rather than just identifying a single new drug, we believe a more comprehensive Systems Therapeutic Approach is needed that includes both pharmacological components (single drug or multi-drug combination) and nonpharmacological components to address the various network imbalances in AD.
Our aim is to identify and develop small molecule compounds as new therapeutics for AD. Our small molecule candidates may be NCEs (New Chemical Entities) or repurposed compounds.
NCEs
- Compounds that have never been tested before
- One of our NCEs is currently undergoing pre-clinical trials in preparation for clinical testing
Repurposed Compounds
- Repurposed compounds are drugs already in use for other conditions.
- They are known to be safe and have well-characterized pharmacological effects
- The allow faster clinical development
- We are testing a repurposed compound in a clinical trial for Mild Cognitive Impairment (MCI), and hope to move toward a clinical trial for AD
Clinical Trials
DDL110
- DDL110 is a new chemical entity (NCE).
- It increases the sAPPα to Aβ ratio and is multifunctional, interacting with two brain cell receptor pathways.
- It is very safe, and enters the brain (passes the blood-brain barrier).
- Improved AD-like deficits in a mouse model of AD to a greater degree than currently approved drugs.
- It is currently moving towards clinical trials for treatement of MCI and AD in the US.
Ongoing Drug Development
The Drug Discovery lab at UCLA is part of the Mary S. Easton Center for Alzheimer's Disease Research and in the Department of Neurology. It is run by experienced scientists, has cutting edge technology, and benefits from collaborations at UCLA and beyond with some of the most pre-eminent research groups in the world.
One of our unique capabilities is compound synthesis. Our ability to rapidly and cleanly synthesize the many compounds we design means we can test and improve these compounds at a rapid pace. We pride ourselves on our use of "Green Chemistry" to make our compounds (3).
The Drug Discovery Lab is first and foremost focused on finding new therapeutics for AD. The field of AD has rapidly expanded in recent years, and a number of physiological systems have been identified to be dysfunctional in AD. We attempt to identify potential drugs that might address imbalances in these systems.
BACE Inhibitors
Amyloid precursor protein (APP) is cleaved by the enzymes BACE and γ-secretase, which leads to the production of Aβ, which aggregates and damages neurons and is thus an important target in AD. Anti-Aβ therapeutics target increased clearance of Aβ from the brain or decrease the production of Aβ. One area of focus in the DDL is decreasing Aβ production by selectively inhibiting the APP cleavage by the enzyme BACE.
- Our goal is to find compounds that only affect the processing of APP by BACE without affecting other molecules processed by BACE.
- We screen compound libraries to identify compounds that decrease BACE activity.
- We improve compounds by making chemical variations (analogs).
sAPPα Enhancers
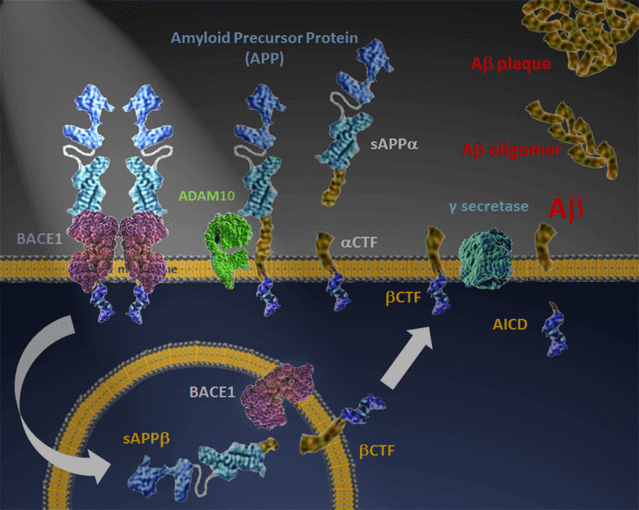
In addition to being cleaved by BACE and γ-secretase to produce Aβ, APP can be processed by an enzyme called ADAM10, which leads to the production of a protein called sAPPα. Most APP is in fact processed by ADAM10. sAPPα is neurotrophic, reduces the production of Aβ, and is needed for the normal functioning of the synapses between neurons.
- Our goal is to find compounds that increase the amount of sAPPα produced in the brain.
- We have already identified a drug candidate that increases sAPPα: DDL110 (an NCE).
- We hope to find many more sAPPα enhancers by screening the 200,000-compound small molecule library at UCLA.
SirT1 Enhancers
Sirtuin 1 is a major longevity determinant that is found in the brain and body, and affects healthy aging.
- SirT1 is found to be lower in the blood serum of MCI and AD patients.
- SirT1 levels are significantly decreased by the major risk factor for AD: ApoE4.
- We want to find compounds that normalize SirT1 levels, especially in more vulnerable individuals with the E4 form of the apolipoprotein gene.
Decreasing Stress Effects
Chronic stress is associated with a higher risk for AD by increasing a chemical modification (phosphorylation) of a protein called tau, which results in ptau.
- Chronic stress is associated with a higher risk for AD.
- It increases ptau and neurofibrillary tangles, hallmarks of AD.
- We have a therapeutic that targets stress pathways and improves cognition in an AD mouse model
- We are improving this compound and will continue pre-clinical testing
Suppression of Exosome-Mediated Tau Propagation Release
An enzyme called neutral Sphingomeylinase2 (nSMase2) is involved in the non-canonical release of exosomes and exosome-mediated tau propagation. Brain permeable inhibitors of this enzyme could suppress the tau pathology in AD models.
- Through screening we have identified potent nSMase2 inhibitors.
- We are evaluating these inhibitors in AD models for suppression of tau propagation.
Inhibiting APP-C31 Production
In addition to BACE/γ secretase and ADAM10 cleavage, APP may be cleaved by an enzyme called a caspase to form a toxic molecule called APP-C31.
- C31 formation is increased during early AD and is associated with inflammation.
- We have identified several small molecules that inhibit the formation of C31, which we will be testing in animal models of AD.
Synthetic Exosomes (SE) for Brain Delivery
It is difficult for most molecules to enter the brain because of the blood brain barrier (BBB). The BBB is created by cells surrounding the blood vessels in the brain and proteins specialized to keep toxic substances out of the vulnerable brain.
- We use pharmacokinetic studies to determine if a compound gets into the brain.
- For molecules that are not brain-penetrant, we have created an SE delivery system using microfluidic synthesis which consists of small deformable nanovesicles (DNV) that can encapsulate drugs and potentially be used to deliver small molecule drugs, protein therapeutics, antibody therapeutics, mRNA and other molecules across the BBB and into the brain.
Window Into the Brain: Exosomes
Exosomes are membranous particles that are secreted by cells and contain proteins cellular components that reflect the physiological state of the cell.
- They can be found in body fluids, such as blood, saliva, and cerebrospinal fluid.
- We have the ability to isolate exosomes that come specifically from the brain.
- We are actively pursuing the use of neuron-derived exosomes to determine the level of alertness and to gain information regarding disease state in neurodegenerative disorders.
- We also hope to develop methods to use exosomes to follow responses to drug treatment.
Clinical Trial in a Dish
This new cutting-edge method can be used to test the effects of therapeutics in differentiated cells derived from pluripotent stem cells originiating from patients with different disease characteristics and genetics.
- Skin cells from patients can be turned into stem cells and then into neurons.
- These stem-cell-derived neurons can then be treated with different therapeutic compounds to test whether cells from different patients respond differently to the same therapeutic.
- Different compounds can be compared for neurons from the same donor.
- We can then tailor clinical trials of these compounds to include only patients whose stem-cell-derived neurons were responsive to the therapeutic. This would be a personalized medicine.
Selected Publications
1 | Duong A, Alam MP, Simmons B, Campagna J, Garg N, John V. Synthesis of Fused Indolines by Interrupted Fischer Indolization in a Microfluidic Reactor. Tetrahedron Lett. 2019 Jan 17;60(3):322-326 |
2 | Campagna J, Spilman P , Jagodznska B, Bai D, Hatami A, Zhu C, Bilousova T, Jun M, Elias CJ, Pham J, Cole G, Jo LaDu M, Jung M, Bredesen D, John V. (2018). A small molecule ApoE4-targeted therapeutic candidate that normalizes sirtuin 1 levels and Improves cognition in an Alzheimer’s disease mouse model. Scientific Reports, accepted 11/18 |
3 | Alam MP, Bilousova T, Spilman P, Vadivel K, Bai D, Elias CJ, Evseenko D, John V. (2018). A Small Molecule Mimetic of the Humanin Peptide as a Candidate for Modulating NMDA-Induced Neurotoxicity. ACS Chem. Neurosci. 9 (3), 462-8. |
4 | Bilousova T, Elias C, Miyoshi E, Alam MP, Zhu C, Campagna J, Vadivel K, Jagodzinska B, Gylys K, John V. (2018). Suppression of tau propagation using an inhibitor that targets the DK-switch of nSMase2. Biochem Biophys Res Commun. 2018 May 23;499(4):751-757 |
5 | Campagna J, Vadivel K, Jagodznska B, Jun M, Bilousova T, Spilman P, John V. (2018). Evaluation of an Allosteric Inhibitor Peptide to identify mimetics that can interact with the Loop F region of BACE and prevent APP cleavage. J Mol Biol. 2018 Apr 10 |
6 | Shkhyan R, Van Handel B, Bogdanov J, Lee S, Yu Y, Scheinberg M, Banks NW, Limfat S, Chernostrik A, Franciozi CE, Alam MP, John V, Wu L, Ferguson GB, Nsair A, Petrigliano FA, Vangsness CT, Vadivel K, Bajaj P, Wang L, Liu NQ, Evseenko D. Drug-induced modulation of gp130 signalling prevents articular cartilage degeneration and promotes repair. Ann Rheum Dis. 2018 May;77(5):760-769 |
7 | Subbiah N, Campagna J, Spilman P, Alam MP, Sharma S, Hokugo A, Nishimura I, John V. Deformable Nanovesicles Synthesized through an Adaptable Microfluidic Platform for Enhanced Localized Transdermal Drug Delivery. Drug Deliv. 2017;2017:4759839 |
8 | Poksay KS, Sheffler DJ, Spilman P, Campagna J, Jagodzinska B, Descamps O, Gorostiza O, Matalis A, Mullenix M, Bredesen DE, Cosford ND, John V. Screening for Small Molecule Inhibitors of Statin-Induced APP C-terminal Toxic Fragment Production. Front Pharmacol. 2017 Feb 15;8:46 |
9 | Heinzelman P, Bilousova T, Campagna J, John V. Nanoscale Extracellular Vesicle Analysis in Alzheimer's Disease Diagnosis and Therapy. Int J Alzheimers Dis. 2016;2016:8053139 |
10 | Alam M.P, Jagodzinska B, Campagna J, Spilman P, John V. C-O bond Formation in a Microfluidic Reactor: High Yield SNAr Substitution of Heteroaryl Chlorides. Tetrahedron Lett. 2016 May 11;57(19):2059-2062 |
11 | Spilman PR, Corset V, Gorostiza O, Poksay KS, Galvan V, Zhang J, Rao R, Peters-Libeu C, Vincelette J, McGeehan A, Dvorak-Ewell M, Beyer J, Campagna J, Bankiewicz K, Mehlen P, John V, Bredesen DE. Netrin-1 Interrupts Amyloid-β Amplification, Increases sAβPPα in vitro and in vivo, and Improves Cognition in a Mouse Model of Alzheimer's Disease. J Alzheimers Dis. 2016 Mar 8;52(1):223-42 |
12 | Peters-Libeu C, Campagna J, Mitsumori M, Poksay KS, Spilman P, Sabogal A, Bredesen DE, John V. sAβPPα is a Potent Endogenous Inhibitor of BACE1. J Alzheimers Dis. 2015;47(3):545-55 |


