Drug Discovery Lab
Drug Discovery Lab
Alzheimer's Disease — a degenerative brain disease that results in progressive memory loss, impaired thinking, disorientation, changes in personality, and severe decline in physical function. Our lab is working on a cure.
Current State of Treatment
Even though more than a century has passed since the first description of Alzheimer's disease (AD), there is no cure for this devastating disease, and the existing approved pharmaceuticals only provide temporary symptomatic relief without slowing down the underlying disease processes. Development of therapeutics for AD has been, arguably, a failure. In the past decade alone, hundreds of clinical trials have been conducted without success, at an aggregate cost of over $1 billion.
In the U.S., there exist only five approved treatments for AD: tacrine, donepezil, rivastigmine, galantamine and memantine. None of these drugs elicit more than unsustained symptomatic improvement, with little or no effect on disease progression. There has been no new drug for AD approved since 2003.
The Drug Discovery Lab (DDL) approach is based on the hypothesis that AD results from an imbalance in an extensive array of networks. Therefore, we are developing Systems Therapeutic Approaches based on known and new targets that can lead to disease modifying therapies for AD.
Alzheimer's Disease (AD)
Alzheimer's disease is an irreversible, progressive brain disorder that slowly destroys memory and thinking skills and, eventually, the ability to carry out the simplest tasks. While most people with Alzheimer's are older than 75, many patients are younger. Alzheimer's disease is the most common cause of dementia among older adults.
The disease is named after Dr. Alois Alzheimer. In 1906, Dr. Alzheimer noticed changes in the brain tissue of a woman who had died of an unusual mental illness. Her symptoms included memory loss, language problems, and unpredictable behavior. After she died, he examined her brain and found many abnormal clumps (called "plaques") and tangled bundles of fibers. These plaques and tangles in the brain are some of the main features of Alzheimer's disease. Another feature is the loss of connections between nerve cells (neurons) in the brain. Neurons transmit messages between different parts of the brain, and from the brain to muscles and organs in the body.
16%
By 2050, this number will double to more than 14 million.
50%
71%
Between 2000 and 2013.
Facts and Figures
As shown above, approximately 16% of persons over 65 (6-7 million people) in the U.S. currently suffer from AD. As the population ages, this number is likely to double to about 14 million people and the cost of care will rise astronomically. But this does not begin to describe the heartbreak for families or burden on caregivers of patients with AD. In fact, caregivers have a higher risk for many diseases than non-caregivers due to the stress of taking care of someone with AD. While the incidence of death due to other diseases such as heart disease or cancer (at a certain age) has declined over the last decade, it has increased for AD, reflecting the limitations for treatment once one is diagnosed with AD (1).
Interestingly, people of different races and ethnic groups have different rates of disease incidence. As shown below, African Americans and Hispanics have a higher rate of AD diagnosis than Caucasians. Identifying the factors that lead to increased risk within a group can help direct the focus of therapeutic development.
Recent News from the DDL
Congratulations to James one of the 2025 Waxman Family Drive Award recipients! This award supports trainees in developing new research ideas related to Alzheimer's and dementia.
Candidate drug that boosts protective brain protein in mice has potential to treat Alzheimer’s Disease
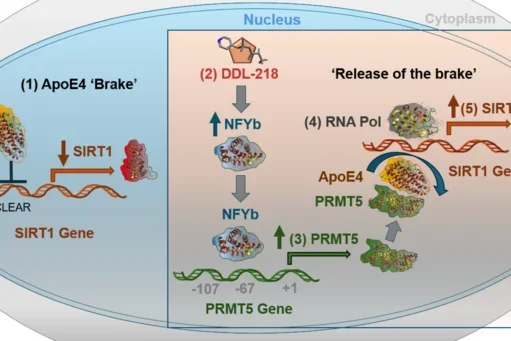
Discovery of an ApoE4-targeted small-molecule SirT1 enhancer for the treatment of Alzheimer’s disease

Celebrating undergraduate graduations
Congratulations Ainsley, Alicia, Angelos, Anisa, Manya, Meghan and Raymond on your graduation! Your hard work and dedication have paid off. Wishing you success and happiness in all your future endeavors!

UCLA researchers develop drug to counteract effects of Alzheimer’s disease, stroke

Award for CNS Delivery of an Enzyme in a Rare Disease
The Amgen Early Innovator Award was presented to the John Drug Discovery Lab (Jesus Campagna & Varghese John) for their project titled "L-Iduronidase in Synthetic Exosomes (SE) for CNS Delivery" at the UCLA Biomedical & Life Science Innovation Day on May 23rd, 2019 by David Piacquad, Senior Vice President of Amgen Business Development.
The award "supports the translation of promising academic research into technologies that benefit patients" and this year's recipients were selected based on "a combination of unmet medical need, technical strength, and competitive differentiation". The lab will use the $75,000 award to further the research on CNS delivery of therapeutic biomolecules.